The bovine Onchocerca ochengi bovine model in African Zebu cattle
1. Biology of Onchocerca ochengi
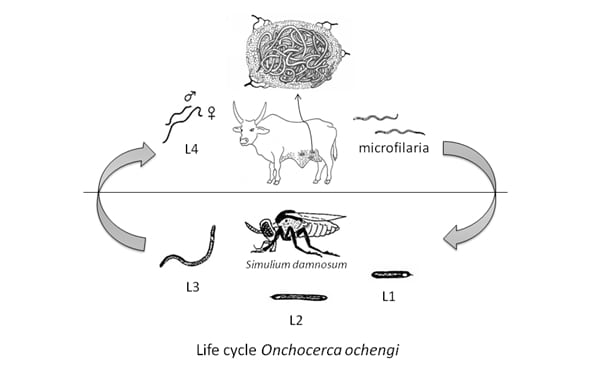
Onchocerca ochengi is a parasitic filarial nematode residing in intradermal nodules (‘onchocercomata’) in the ventral skin of African cattle. It is the closest known relative of O. volvulus, the causative agent of river blindness in humans, with which is shares the same blackfly vector, Simulium damnosum s.l.. The phylogenetic closeness of the two filarial species, the same vector and their geographic co-existence in the savanna of Northern Cameroon prompted us to develop, in co-operation with colleagues from the Liverpool School of Tropical Medicine and the Institut de Recherches Agronomiques pour le Developpement at Wakwa, this easy-to-access bovine filaria to become the bovine O. ochengi model. In our DFG-COBE project, we actually keep a herd of 60 cattle close to a Simulium breeding site along the river Vina du Sud near Ngaoundéré. In these study animals, kept exposed to natural transmission, we follow-up the acquisition of nodules and O. ochengi microfilariae since up to 5 years by now (2015).
In the vector, after ingestion of the microfilariae by a bloodmeal on the cattle host, the filaria develops within the flight-muscles of the fly. After 6 to 8 days and two moults, the third-stage larvae are ready, and they migrate to the head or thorax of the blackflies.
Life cycle of Onchocerca ochengi in cattle.
Ventral skin of a highly parasitized zebu cow from Cameroon with many Onchocerca ochengi nodules. These animals, though, do not suffer from any clinical signs due to infection. Copyright 2014 Programme Onchocercoses.
Most of the Simulium flies take their bloodmeal in the morning and/or afternoon from the inguinal region of cattle. This is also where most nodules can be found (see photo). Occasionally, they appear on the hind legs, buttocks, front leg armpits, neck, ears and face near the eyes. These are also the regions where the vast majority of microfilariae occur. Only very rarely nodules are found on the chest and back.
Besides O. ochengi sensu stricto, another haplotype O. ochengi ‘Siisa’ has been discovered in Cameroon (Eisenbarth et al. 2013; Hildebrandt et al. 2014). This type has originally been described from a single developing larva, found in a blackfly caught near the Siisa river in Uganda (Krüger et al. 2007). That means they must have been two geographically isolated populations, which are mingling again in Cameroon. There seem to be also morphological differences of the infective larvae among these populations (unpublished results). Most interestingly, according to mitochondrial markers this new haplotype is phylogenetically as distant to O. ochengi s.s. as to O. volvulus, showing their close evolutionary connection. It is believed that the human parasite O. volvulus originated from a spillover event of a common ancestor with O. ochengi from cattle. That must have happened during the domestication of cattle between 5,000 and 10,000 years ago.
Population studies of O. ochengi in single cattle host individuals revealed that the sedentary females copulate and produce offspring with different males, which can migrate between nodules (Hildebrandt et al. 2012, 2014). A large proportion of males, however, stay in the female’s nodule, on average 1 female with 1 male. Another very interesting finding was that the density of microfilariae in the skin does not correlate with the number or location of adult worms, indicating some sort of density-dependent control of the parasite.
Literature: => see publications for our own papers
Eisenbarth et al. 2013
Eisenbarth et al. (in preparation)
Hildebrandt et al. 2012
Krueger, A., P. Fischer, et al. (2007). “Molecular phylogeny of the filaria genus Onchocerca with special emphasis on Afrotropical human and bovine parasites.” Acta Trop 101(1): 1-14.
Hildebrandt et al. 2014
Renz et al. 1994
Wahl et al. 1991
Wahl et al. 1994
2. Immunology
Although O. ochengi is the closest known relative to O. volvulus, it does not inflict pathology in its natural host. Cattle differ in their susceptibility to parasite acquisition, on the adult worm stage (i.e. number of palpable nodules), microfilarial stage (i.e. microfilarial densities in the skin), and on microfilarial stages of other filarial species, such as O. dukei, O. gutturosa, O. armillata, Certopithifilaria spec., Setaria spec. and possibly more. More susceptible animals showed a higher humoral immune response than more resistant ones (Manchang et al. 2013).
Cross-protecting vaccination has been demonstrated by inoculating live O. volvulus infective larvae to naive Zebu cattle. These animals were highly protected from O. ochengi infection as compared to a mock-treated control (Achukwi et al. 2007).
Recently, the antimicrobial activity of some Onchocerca peptides was described from excretory-secretory products, isolated from in vitro-cultured O. ochengi adult females (Eberle et al. 2014). If this finding can be confirmed under physiological conditions in vivo, it may hint to a protective effect of filarial nematode infections against microbial pathogens.
Literature:
Brattig et al. 2000
Achukwi et al. 2004
Achukwi et al. 2007
Manchang et al. 2013
Eberle et al. 2015
3. Search for macrofilaricidal drugs
Because the biology of O. ochengi is overlapping with O. volvulus excluding its pathologic effects, it is an excellent model for the screening of chemotherapeutic agents for their filaricidal activity. Just as in humans, ivermectin neutralizes all microfilariae over a short time with only moderate side effects in the host. In fact, the drug stems from its widespread use in veterinary medicine and was later adopted for human mass treatment. However, efficient compounds against the adult stage are still not available for treating endemic populations at risk. Several compounds have been tested, in co-operation with IRAD Wakwa and the Liverpool School of Tropical Medicine for their macrofilaricidal activity in cattle, like Suramin and CGP20376 (Renz et al. 1995), Tetracycline (Langworthy et al. 2000), UMF078 (Broonsvoort et al. 2008) and repeated high doses of ivermectin (Broonsvoort et al. 2005).
A final breakthrough has not been yet reached, although most of the tested compounds showed macrofilaricidal activity. However, often the compounds were also fairly toxic for the host, or the required treatment regimen was not practical for large-scale treatment intervention programs.
In particular, the discovery that neutralization of the symbiotic Wolbachia bacteria by tetracyclines kills the adult worm lead to successful trials for humans in areas where onchocerciasis is co-endemic with loasis, another filarial-borne disease, which causes severe, sometimes fatal adverse events after ivermectin treatment in the affected population.
Literature:
Renz et al. 1995
Cross et al. 1997
Langworthy et al. 2000
Gilbert et al. 2005
Broonsvoort et al. 2005
Broonsvoort et al. 2008
Husna et al. 2010
4. Vector and transmission
Quantitative entomological data of longitudinal studies of vector species and abundance, biting frequencies, Onchocerca spp. infection rate and L3 transmission potential are important epidemiological data needed for risk assessment and management of control strategies. Thus, these parameters have been determined by entomological studies since 1987. Simulium damnosum blackflies are collected at fly-catching sites near the breeding rivers and subsequently dissected under the microscope for the presence of filarial larvae.
Simulium damnosum transmits filariae of human and of animal origin. In Cameroon, at least three different species are sympatric: O. volvulus, O. ochengi (s.s. and ‘Siisa’) and O. ramachandrini, a filaria from the warthog. The species seasonality can be different, as seen by the higher prevalence of O. ramachandrini in the rainy season, whereas O. volvulus and O. ochengi transmission is higher during the dry season in North Cameroon.
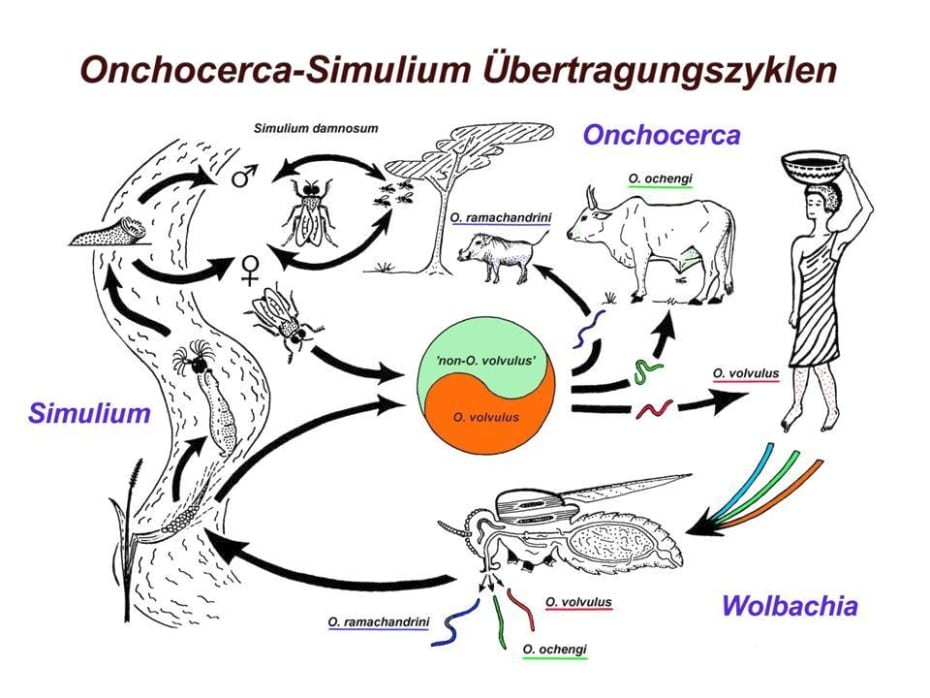
Cross-transmission cycles of human and animal Onchocerca spp. by Simulium damnosum s.l. in North Cameroon (A. Renz)
For species differentiation of the third-larval stage, the body length and shape of anterior/posterior endings can be used (Renz 1987, Wahl et al. 1994). Generally, O. ramachandrini reach a longer body length than O. ochengi, and O. volvulus has the shortest mean body length. The problem is that the body size distribution is overlapping between species, making alternative identification methods desirable. In the past, DNA probes were generated to identify O. volvulus from non-volvulus (Seidenfaden et al. 2001). Nowadays, PCR-based methods employing conserved regions of mitochondrial DNA facilitate the identification of the species, even for unknown filarial nematodes, and the elucidation of their phylogenetic relationships (Eisenbarth et al. 2013).
Literature:
Renz 1987a,b
Renz et al.1987a,b,c
Wahl et al. 1994
Seidenfaden et al. 200i
Basanez et al. 2007
Eisenbarth et al. 2013
Eisenbarth et al. (in preparation)
5. Zooprophylaxis
On the Adamaoua plateau, where cattle stocking density is very high throughout the year, onchocerciasis prevalences in the human population are generally lower than in regions with lower cattle stock densities (Renz et al. 1994, Wahl et al. 1998). Two hypotheses have been formulated to explain this observation: Zooprophylaxis, meaning that a proportion of the vector population is diverted to bite on cattle and other vertebrate hosts, thereby reducing the biting rates in humans.
And crossprotective premunition by O. ochengi L3 transmitted onto man. Premunition means that after an O. ochengi-infected blackfly (and possibly with other non-volvulus filariae) bites a human host, the invading L3 trigger a humoral immune reaction, which is cross-reacting with O. volvulus antigens, therefore protecting from an infection. Experiments with cattle, crossprotected by heterologous immunization with live O. volvulus L3, have supported this hypothesis (Achukwi et al. 2002)
Literature:
Achukwi et al. 2002
Renz et al. 1994
Wahl et al. 1998