The current project, titled “Analysis of host-parasite cross-talk based on the bovine model for human onchocerciasis, Onchocerca ochengi” (DFG1) focuses on the study of the population biology of Onchocerca filarial nematodes in cattle by using state-of-the-art molecular approaches to improve understanding of the role of the secretome of O. ochengi in naturally infected Zebu-cattle. Using new epidemiological data, the project aims to develop sustainable strategies for the control of human onchocerciasis in the Republic of Cameroon.

DFG1 Study aims:
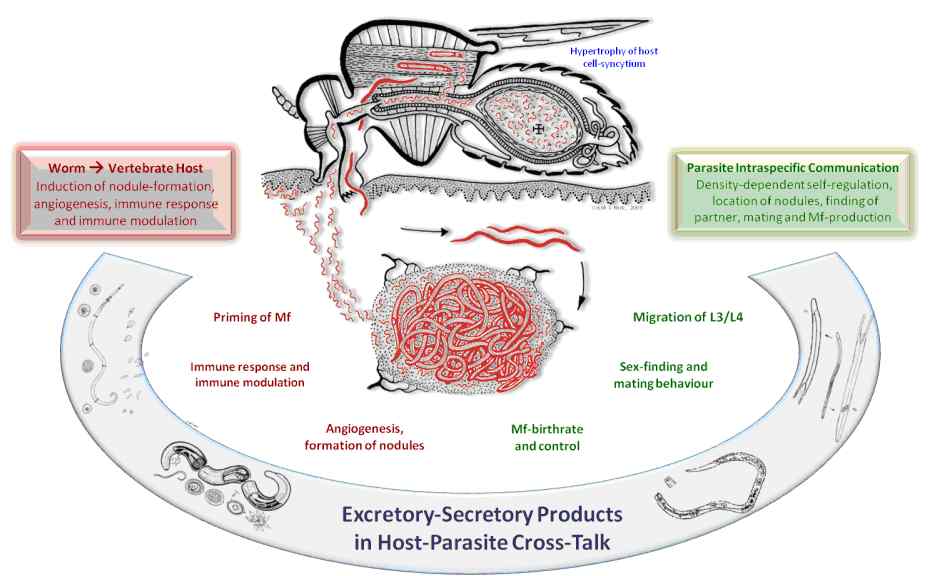
• To target the analysis of the secretome of O. ochengi of which adult reproducing stages and larval stages will be subjected to production of E/S- products, with subsequent proteomic identification of the major secreted proteins, thereby possibly revealing unexpected and unknown proteins which may contribute to the helminth-induced immune modulation.
• Following sequence analysis, full-length ORFs will be isolated and proteins will be recombinantly expressed using both, prokaryotic and eukaryotic expression systems. Following purification, these target molecules will be examined by complementary analytical methods.
1. Candidate proteins have been selected (incl. proteases, lectins, cytokine homologue, retinol-binding proteins and proteins with unknown functions) that will be tested for their immune modulating functions in vitro and in vivo, employing murine model antigen systems for T and B cell responses. The involvement of candidate proteins in a protective immune response will be tested employing the murine system of S. ratti infection and O. ochengi infected cattle.
2. To determine tissue- and stage-specific distribution, candidate proteins will be analysed using RT-PCR and immunohistochemistry.
3. Comparative studies of putative orthologs will be performed using C. elegans, allowing transgenic approaches to unravel novel physiological functions.
4. Immunogenic proteins have been identified in previous studies. Besides their “visibility” to the immune system, their biological functions are largely unknown. The structure-function relationship of selected proteins will be analysed. The detailed knowledge of their 3-D structure will help solve their molecular function.
The most prominent feature of the bovine model used, is the unique fact, that all stages in the life cycle of the parasite can be quantitatively assessed, both in the vector and in the vertebrate host. Therefore, it is possible to study, by cross-sectional and longitudinal studies in herds of cattle exposed to natural transmission, the density-dependent factors that govern the numbers and reproduction success of individual worm-populations in individual cattle.
• We propose to address the basis parameters of host-parasite population dynamics by a careful longitudinal follow-up study of a herd of local Gudali-cattle, kept from their birth on at a pasture with high S. damnosum biting rate and O. ochengi transmission, focussing on the factors which govern the acquisition and turn-over of the parasite-population in the host.
• The parasitological and entomological data collected will be used to develop mathematical models urgently needed to assess and predict the outcome of various control strategies for human Onchocerciasis.
This project will lead up to 2017. As a part of the entomological monitoring we are assessing the dynamics of Simulium vector populations and the residual Onchocerca volvulustransmission potentials in the area of Touboro, Tchollire and Ngaoundere. At these sites our research teams have already obtained reliable pre-control data of ABRs and ATPs.
In addition, molecular tools will be used to distinguish the three species. Our research will also focus on important individual measures of protection against the bites of the vector flies. Exposure generaly decreases with rural development and better clothing; however, this has to be confirmed by direct observation and must be measured quantitatively (co-operation with male & female anthropologists from the Univ. of Ngaoundere/Buea).
For the collaborators in this project, see PARTNERS. More information on this collaboration will shortly be available online and can be found under: www.uni-muenster-cameroon.de


Leave a Reply
Want to join the discussion?Feel free to contribute!